
This MedCase describes treatments for female sexual dysfunction, reviewing recent evidence for the safety and efficacy of testosterone, with guidance on prescribing in general practice.
Mrs R is a 58-year-old postmenopausal woman who presents with concerns about her libido, which has reduced gradually over the past ten years. She has always had a good relationship with her husband, but lately her lack of interest in intimacy has begun to bother them both.
Mrs R went through menopause at age 53 and is stable on menopause hormone therapy (MHT). During the perimenopause, she started estrogen and cyclical progesterone to improve her symptoms of hot flushes, poor sleep and low mood. Once her bleeding stopped, she switched to continuous use, which she tolerates well and gives good symptom control.
Her sleep is mainly good, and she has no symptoms of anxiety or depression. However, she tells you she worries about the effect of the lack of sexual intimacy on her relationship.
She wonders if menopause has made it worse.
How do you approach this consultation?
Low libido: When is it a problem?
Female sexual dysfunction reflects a complex, interacting mix of biological, psychological, relational, and sociocultural determinants: hormonal and medical conditions, pain and medication effects, mood and stress-related factors, partner and communication dynamics, and broader social influences such as body image and cultural norms. These factors commonly overlap, and it is the cumulative interplay, rather than any single driver, that underpins symptoms and impacts sexual wellbeing.
A thorough assessment should identify potential confounders of low desire, including medications (such as antidepressants and antihypertensives), significant life events (pregnancy, lactation, surgery), and relationship stressors. While fluctuations in libido across life stages are normal and often benign, changes in sexual desire can still meaningfully affect quality of life. These concerns should be explored in a patient-centred, non-dismissive manner, focusing on how symptoms impact daily functioning, relationships, and overall wellbeing.
In the DSM-IV, hypoactive sexual desire disorder (HSDD) was defined as:
“Persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity with marked distress or interpersonal difficulty not otherwise accounted for by a general medical or psychiatric condition”.
DSM-5 eliminated the term HSDD as a standalone diagnosis and combined desire and arousal problems into a single diagnosis: Female Sexual Interest/Arousal Disorder (FSIAD).
The diagnosis of FSIAD in clinical practice should be based on a thorough clinical assessment guided by diagnostic criteria.
For women with acquired, generalised FSIAD, a targeted physical and gynaecological examination, and additional laboratory testing when indicated (prolactin, thyroid function, estradiol, progesterone, LH), may help identify modifiable contributors.
Management should follow a biopsychosocial model, combining pharmacological (hormonal and non-hormonal), psychological, and behavioural interventions as appropriate. Therapeutic intervention with transdermal testosterone should only be initiated in women after correction of modifiable biopsychosocial factors. Transdermal testosterone targeting central and neuroendocrine pathways has demonstrated efficacy for postmenopausal FSIAD.
The International Society for the Study of Women’s Sexual Health (ISSWSH) 2021 guidelines1 endorse transdermal testosterone therapy for postmenopausal women with hypoactive sexual desire disorder. While the 2019 Global Consensus Position Statement formally recommended use only in postmenopausal women, the 2021 ISSWSH guidance notes limited evidence supporting cautious use in late-reproductive-age premenopausal women.
Transdermal testosterone may therefore be considered for postmenopausal women whose FSIAD is not primarily driven by modifiable factors or comorbidities such as relationship issues or mental health conditions. Current evidence demonstrates a moderate therapeutic benefit, and safety data related to short-term use at physiological doses are reassuring. Long-term safety remains uncertain. Initiation should occur only after a clear informed-consent discussion.
Shared decision-making requires a transparent discussion of expected benefits, limitations, and potential risks, including unknown long-term risks.
Total testosterone should be measured at baseline and periodically thereafter, solely for monitoring to ensure levels remain within the physiological premenopausal range; testosterone levels must not be used to diagnose FSIAD. Compounded formulations are not recommended due to a lack of sufficient evidence for their efficacy and safety.
Assessment
Libido may be a sensitive topic and patients can be reluctant to raise it.
A useful screening question is: Are there sexual concerns you wish to discuss?
Other questions may include:
- Do you have dryness, discomfort or pain during vaginal penetration?
- Have you experienced a loss of sexual desire and sexual thoughts?
- Have you found you are less responsive to your partner sexually?
- Are you less responsive to sexual stimulation?
- Do you have difficulty reaching orgasm?
- If your answer is yes to any of the above, does this bother or distress you?
- If your answer is yes to any of the above, would you like treatment to change things?1
The Decreased Sexual Desire Screener (DSDS)2 is a validated tool suitable for clinicians diagnosing low sexual desire in women. It helps identify women who warrant further assessment. Follow with a comprehensive history, examination, and appropriate investigations (including medical, psychological, relationship, and cultural factors) if the screening is positive. A positive DSDS is not a definitive diagnosis, clinical judgment is required.
Screening questions:
- In the past, was your level of sexual desire or interest good and satisfying to you?
- Has there been a decrease in your level of sexual desire or interest?
- Are you bothered by your decreased level of sexual desire or interest?
- Would you like your level of sexual desire or interest to increase?
- Please circle all the factors you feel may be contributing to your current decrease in sexual desire or interest:
- Medications, drugs, or alcohol you are currently taking
- An operation, depression, injuries, or other medical condition
- Pregnancy, recent childbirth, or menopausal symptoms
- Other sexual issues you may be having (pain, decreased arousal, or orgasm)
- Your partner’s sexual problems
- Dissatisfaction with your relationship or partner
- Stress or fatigue.
Sexual history taking should explore both partnered and unpartnered sexual activity, as well as past and current relationships and sexual experiences. When there is a discrepancy in desire between the patient and her partner, a diagnosis of FSIAD should only be made if this discrepancy results in distress for the patient herself.
Note: the DSDS was validated in pre-menopausal women with acquired/generalised low desire. Caution applying it to post-menopausal women, situational desire issues, or when arousal/other domains dominate.2
DSM-5
According to DSM-5, symptoms must include ≥3 of the following, persisting for ≥6 months, causing clinically significant distress, and not better explained by another mental/medical/relationship condition or medication/substance:
- Interest/arousal symptoms (need ≥3):
- Absent or reduced sexual interest.
- Absent or reduced sexual/erotic thoughts or fantasies.
- Reduced initiation of sexual activity, and typically unreceptive to partner’s attempts.
- Reduced sexual excitement/pleasure during sexual activity in ≥75% of encounters.
- Reduced sexual interest/arousal in response to sexual cues or erotic stimuli.
- Reduced genital or non-genital sensations during sexual activity in ≥75% of encounters.
- Duration
- Symptoms present for ≥6 months.
- Distress
- Must cause clinically significant distress.
- Not otherwise attributable to:
- Severe relationship distress, domestic violence
- Other psychiatric disorders
- Substance/medication effects
- Medical conditions.
Patients may find it helpful to understand more about female sexual desire and arousal, and there are some good resources available from:
- The Jean Hailes women’s health website.
- The book “Where Did My Libido Go?” by Dr Rosie King.
- The Australasian Menopause Society (AMS) information sheet on Sexual Difficulties in the Menopause.
Physiology: What role do hormones play?
The physiology of sexual desire and interest is complex and individual; hormones are only one component. Factors such as relationship issues, health comorbidities (e.g. diabetes), previous sexual or emotional abuse, and body image issues can impact a woman’s desire for sex. It is important to elicit non-hormonal factors contributing to low libido, as these are targets for therapy.
Estrogen levels are directly related to libido, as evidenced by studies showing a correlation between reduced estrogen and reduced sexual function3. This is particularly relevant during the menopausal transition, when declining estrogen causes vaginal dryness and can make intercourse uncomfortable. Other menopause-related sleep and mood issues may also contribute to reduced libido and overall energy. Further details on the evaluation and management of genitourinary symptoms of menopause are available in this Goodfellow Unit MedCase.
Testosterone in women is produced by the ovaries and adrenal glands, with around half derived from peripheral conversion of androgen precursors. Levels decline gradually with age and fall sharply after bilateral oophorectomy. Androgen decline in postmenopausal women relates primarily to age-related ovarian and adrenal changes rather than the menopausal transition itself. Although very low DHEA-S levels correlate modestly with reduced sexual responsiveness in older women, most women with low DHEA-S have no sexual dysfunction.
There is no absolute testosterone threshold that defines FSIAD, reflecting the complexity of androgen metabolism and the poor correlation between circulating levels, receptor interaction, and tissue concentrations1.
The ISSWSH process of care for management of hypoactive sexual desire disorder (HSDD) in women
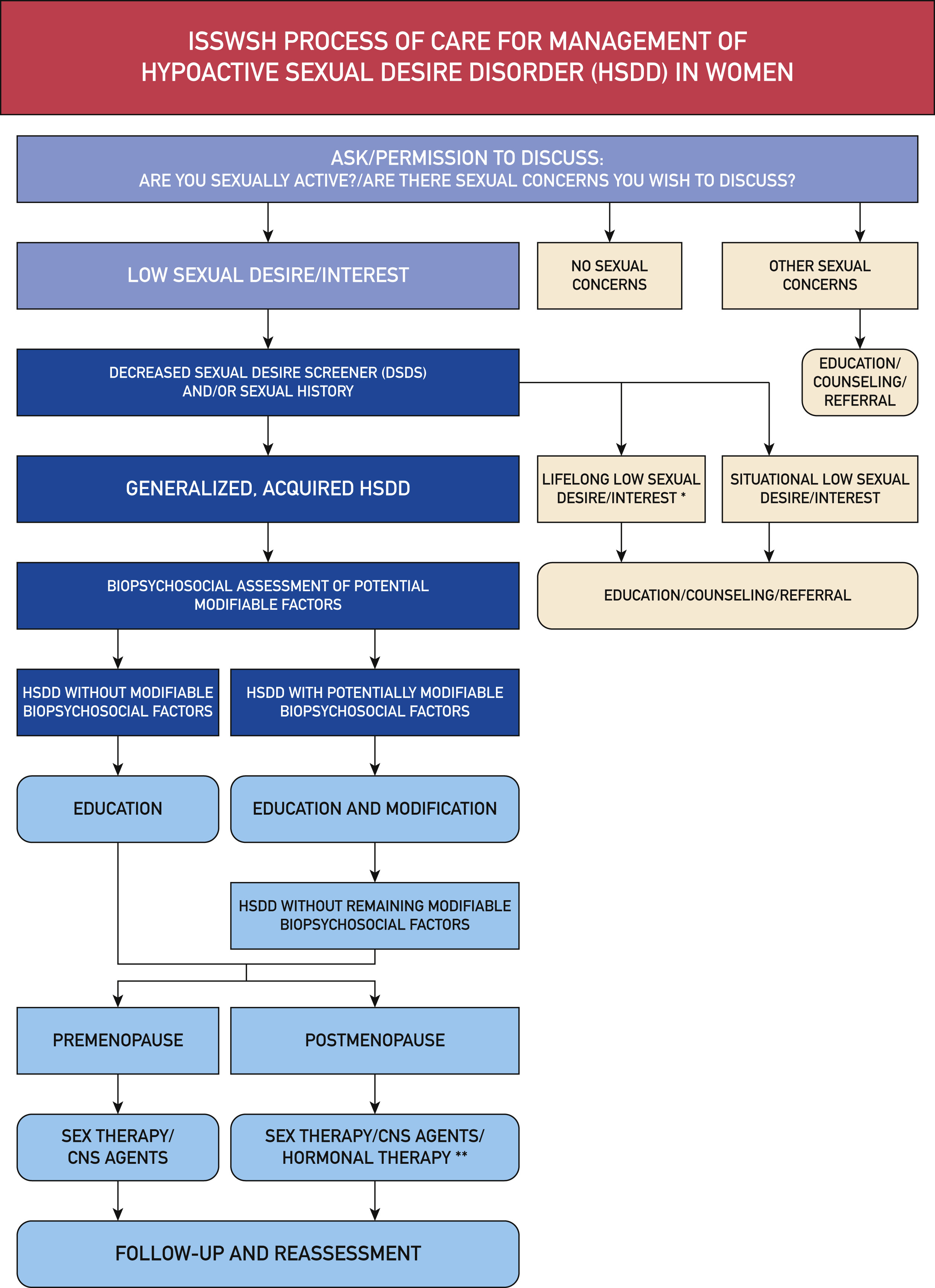
What therapies are available?
Therapies for reduced libido target the multifactorial causes and must be individualised to best target the key contributing factors for each patient.
- Treatment of genitourinary symptoms of menopause (GSM) to minimise dyspareunia (see the Genitourinary syndrome of menopause Goodfellow MedCase)
- Treatment of anxiety or depression if present.
- Relationship counselling.
- Cognitive-behavioural therapy or other psychological intervention from an appropriately qualified therapist (see Sex Therapy NZ website).
You discuss libido further with Mrs R.
She cannot identify any particular relationship or body image issues affecting her libido.
She and her husband saw a counsellor last year, who recommended some behavioural strategies, but these have made little difference. Her MHT dose is stable.
Could testosterone be a suitable treatment for Mrs R?
Transdermal testosterone therapy
AndroFeme® 1 was MedSafe approved in New Zealand in April 2025. It is indicated for the treatment of hypoactive sexual desire disorder (HSDD) in postmenopausal women4. Using this product outside of the approved indication, eg, in perimenopause, is an unapproved indication which requires appropriate consent.
Treatment should only be commenced after appropriate education and optimisation of modifiable biopsychosocial contributors.
Dosing
- Start: 5 mg testosterone (0.5 mL) once daily, applied at the same time each day to the upper outer thigh or buttock.
- Titrate: If symptoms have not improved after 3 months and total testosterone remains within the premenopausal physiological range, the dose may be increased to 10 mg (1 mL) daily.
- Expected response: Improvements in sexual motivation typically emerge after 4 - 8 weeks.
- Stop: If no meaningful improvement is seen after 6 months, discontinue therapy and consider alternative approaches.
- Prescribing: Not funded by Pharmac (at time of writing).
- Technique: Patients should use the graduated applicator to measure the correct dose and apply it immediately to clean, dry skin on the upper outer thigh or buttock. The cream should be massaged in evenly until fully absorbed (usually ~30 seconds), and hands should be washed with soap and water afterwards. The applicator may be rinsed in hot water after use. Absorption may be less predictable if applied to other body sites, and it should not be applied to the genital or perineal area. Patients should avoid bathing, showering, swimming, or applying cosmetics or sunscreen to the application site for at least four hours.
Monitoring
A baseline total testosterone and sex hormone-binding globulin (SHBG) level should be obtained prior to starting therapy, with repeat measurements at 3–6 weeks and then every 6 months thereafter.
It is recommended that women should ideally attend the same laboratory for baseline testosterone biochemistry prior to and during treatment.
Serum total testosterone should be kept within the physiological premenopausal range as determined by the local laboratory reference. This should be specified on the lab request.
Testosterone levels should guide clinical decision-making rather than determine efficacy, as serum concentrations do not correlate with treatment response. There is no target therapeutic level. Testing is primarily used to:
- Exclude women with mid-range or high baseline values, which makes an androgen-related cause for symptoms unlikely; and
- Monitor therapy to ensure levels do not become supraphysiological, thereby reducing the risk of androgen-excess side effects1.
The primary measure of benefit is the woman’s subjective improvement in sexual function.
If levels exceed the upper limit of the assay’s premenopausal range, assess for signs of hyperandrogenism and consider reducing the dose. Women with levels > 50% above the upper limit should be advised to decrease the applied dose.
A review at 12 weeks should include a comprehensive assessment of efficacy and safety. A daily dose of up to 1mL (10 mg) is usually sufficient and should rarely need to be exceeded. If there is no clinical benefit by 6 months, treatment should be discontinued.
Testosterone therapy can cause androgenic effects such as acne, hirsutism, voice deepening, and androgenic alopecia. If serum testosterone becomes supraphysiological, the dose should be reduced, even in the absence of androgenic symptoms, with repeat testing after 2–3 weeks.
Once stable, serum testosterone should be monitored every 4 - 6 months to detect inadvertent overuse. Patients should be routinely assessed for signs of androgen excess, including acne/oily skin, increased facial hair, and scalp hair thinning. If androgenic side effects occur, the dose should usually be decreased. Notably, side effects may occasionally develop despite standard dosing or normal serum levels.
Baseline liver function tests and a lipid profile are recommended prior to initiation of treatment. Although physiologic doses of transdermal testosterone do not typically alter these parameters, underlying liver disease or significant hyperlipidaemia are contraindications to treatment1.
Earlier guidelines recommended annual breast and pelvic examinations, mammography, monitoring for abnormal bleeding, and periodic testing for lipid levels, liver function, and a complete blood count.
Based on current safety data, it is reasonable to follow national screening recommendations, including mammography and cardiovascular risk assessment1.
Recommended monitoring for testosterone therapy in women
| Before initiation (baseline) | |
|
Investigations Ensure use of same laboratory for serial tests. Specify premenopausal female reference range on lab form. Women with mid-range or high baseline testosterone (androgen-related cause unlikely). Establish baseline for safe longitudinal monitoring. |
Total testosterone SHBG LFT Lipid profile |
| Symptoms | Document sexual symptoms + degree of distress. |
| Identify contraindications |
| 3-6 weeks after starting | |
|
Investigations Ensure serum levels remain within local premenopausal physiological range. |
Total testosterone |
| Symptoms |
Assess subjective improvement in sexual function. Review for androgenic symptoms (acne, oily skin, hirsutism, scalp hair thinning). Treatment response is clinical, not biochemical - no target level. |
| Identify contraindications |
| 12 week review | |
|
Investigations If levels > upper limit of normal: assess for hyperandrogenism and reduce dose. If >50% above upper limit: advise dose reduction immediately. |
Total testosterone |
|
Symptoms Clinical benefit should be evident by 12 weeks. |
Full assessment of efficacy and safety. Review side effects and dose. Evaluate need for dose adjustment. |
| Identify contraindications |
| Ongoing (4-6 monthly once stable) | |
|
Investigations
|
Total testosterone Cardiovascular risk and other age-appropriate screening assessments per national guidelines and individual risk factors. |
|
Symptoms Clinical benefit should be evident by 12 weeks. Androgenic effects may occur even with normal serum levels; dose reduction is usually required. If testosterone becomes supraphysiological, repeat testosterone in 2 - 3 weeks. |
Review for androgen excess (acne, hirsutism, alopecia, voice changes). Ongoing sexual function assessment. |
| Cessation |
If no benefit by 6 months, discontinue therapy. Lack of subjective improvement indicates therapy is ineffective. |
Routine general health monitoring
- Age-appropriate breast and pelvic examinations
- Mammography per national screening recommendations
- Routine cardiovascular and metabolic screening. Earlier guidelines recommended annual LFTs/lipids/FBC, but current evidence supports standard national screening pathways and unless clinically indicated.

Contraindications and Precautions
Contraindications
AndroFeme® 1 should not be used in women with:
- known hypersensitivity to testosterone
- known sensitivity/allergy to tree nuts (almond oil), or any excipients
- known or suspected breast cancer
- known or suspected androgen-dependent neoplasia
- nephrotic syndrome
- history of thromboembolism (due to lack of data)
- hypercalcaemia
- pregnancy or lactation
- normal reproductive function without reliable contraception, due to the risk of virilisation of a female foetus.
Precautions
- Anti-doping: High-performance athletes must be aware of relevant rules regarding androgen use.
- Transference risk: Although the dose is low, testosterone can be transferred by close skin-to-skin contact.
- Avoid contact between the application site and partners/children.
- Cover the application area with clothing once dry.
- Washing with soap and water removes most residual testosterone.
- Long-term close contact with young children may result in passive absorption and potential virilisation.
- Cancer risks:
- Use cautiously in women at risk of or with endometrial hyperplasia/cancer.
- Long-term effects on ovarian cancer risk are unknown; use cautiously in women at risk or with current disease.
- Concomitant oestrogen therapy: Not recommended with conjugated equine oestrogens (CEE), as clinical studies showed no significant improvement in sexual function when combined with transdermal testosterone.
Adverse effects
Common adverse events (≥1/100 to <1/10) reported in clinical trials
| Adverse event | Testosterone | Placebo |
| Acne | 122 (7.5%) | 83 (5.0%) |
| Increased hair growth | 212 (8.6%) | 106 (6.1%) |
| Alopecia | 55 (4.5%) | 55 (4.4%) |
| Voice change | 48 (3.7%) | 44 (3.4%) |
Post-marketing reports: thinning of hair and dizziness. In clinical trials, postmenopausal women using a transdermal testosterone patch for three years experienced a small mean weight increase of 1.52 kg1,4.
Evidence: Does transdermal testosterone therapy work?
There is consistent evidence from placebo-controlled RCTs that testosterone therapy is more effective than placebo for treating FSIAD in postmenopausal women.
Transdermal testosterone improves sexual desire and reduces sexually related distress in both naturally and surgically menopausal women, regardless of whether they are taking estrogen ± progestogen therapy.
Across studies, additional benefits include increased frequency of satisfying sexual events, enhanced arousal, orgasm, pleasure, responsiveness, and improved sexual self-image, with reductions in sexual concerns. Clinical benefit is most evident when serum testosterone levels are maintained within the physiologic premenopausal range1.
Evidence in premenopausal women is more limited. Three placebo-controlled trials in late-reproductive-age women suggest potential benefit, but sample sizes were small, and a recent meta-analysis concludes that further research is needed before efficacy can be confirmed1.
Long-term safety data are limited.
The longest placebo-controlled study followed 331 women for 24 months. A meta-analysis of 36 trials (8,480 women; duration 12 weeks to 2 years) found increased risks of acne (RR 1.46; 95% CI 1.11–1.92) and hair growth (RR 1.69; 95% CI 1.33–2.14)5. No significant improvements were noted in bone density, body composition, or cognition, although a small increase in weight was observed. Lipid effects were confined to oral preparations, which are no longer recommended because of hepatic toxicity.
Most research has examined transdermal patches delivering 150–300 mcg/day. A meta-analysis of seven trials (3,035 participants) using 300 mcg/day for 24 weeks showed higher rates of androgenic adverse effects (HR 1.37; 95% CI 1.12–1.69), including acne (HR 1.41) and hair growth (HR 1.56), without alopecia, clitoromegaly, or voice deepening. No significant differences were seen in lipid profiles, glucose metabolism, cardiometabolic markers, renal or liver function6. Small studies found no change in mammographic breast density, but data are insufficient to assess long-term breast cancer risk.
Safety data at physiological doses beyond 24 months are lacking. A large phase-3 cardiovascular and breast safety trial of 1% testosterone gel (300 mcg daily) enrolled 3,656 postmenopausal women with HSDD and at least two cardiovascular risk factors. Over four years (>7,300 woman-years), 53 adjudicated cardiovascular events were reported - fewer than expected - and breast cancer rates were consistent with age-related background incidence7.
Despite these reassuring findings, long-term safety - particularly regarding cardiovascular events (MI, stroke, VTE), breast cancer, endometrial and ovarian effects, cognition, mood, and musculoskeletal health remains uncertain. Existing trials have been neither large enough nor long enough to define long-term risks with confidence1.
| Key findings from the testosterone meta-analysis5 |
|
In post-menopausal women:
|
You suggest that transdermal testosterone in the form of AndroFeme® 1 may be an option.
You note that Mrs R does not have hyperlipidaemia and her cardiovascular risk is low. She is up to date with all screening, and there is no history of liver disease.
After a detailed conversation regarding risks, benefits, the lack of long-term safety data, costs, and Section 29 status, you arrange baseline testing with an agreement to issue a prescription after the results are received.
You ask her to monitor her symptoms as well as any side effects. A total testosterone level is arranged for 6 weeks and at 12 weeks, with reviews at this stage.
After three months, her total testosterone is within the local laboratory’s normal reference range for pre-menopausal women.
Mrs R reports having more energy and says she is enjoying sex again, with a noticeable improvement in libido. There are no symptoms or signs of androgen excess.
You agree to continue the cream at the current dose and with regular 3-monthly reviews of bloods and symptom monitoring.
This MedCase was created by Dr Danuta Amelung, BHB, MBChB, FRNZCGP, AFRACMA, Dip. Paed, PGCertWHlth, PGDipTrvMed.
References
Supported with an unrestricted educational grant from
Recognition of Learning Activities
Don't forget to log your time with The Royal New Zealand College of General Practitioners portal for recognition of learning activities.



