
Empagliflozin (brand name: Jardiance) is a sodium glucose co-transporter 2 (SGLT2) inhibitor with important benefits for patients with type 2 diabetes and concomitant heart failure, cardiovascular disease (CVD) or renal disease.1
This MedCase considers the place of empagliflozin alongside existing treatments and describes how to safely start treatment.
Mr V is a 62-year-old Tongan minister with type 2 diabetes who has been on insulin for 15 years. He has diabetic retinopathy and albuminuria and is obese at 107kg, with a history of hypercholesterolaemia, hypertension and non-alcoholic fatty liver disease.
His last HbA1c was 84 mmol/mol and his current medications are:
- Metformin 1000mg BD
- Glipizide 5mg BD
- Novomix 30 Flexpen 90 units BD (insulin aspart 30 IU/mL + insulin aspart protamine 70 IU/mL)
- Losartan 50mg daily
- Felodipine 10mg daily
- Atorvastatin 40mg daily
Mr V was previously under the care of the hospital team but did not attend his last three clinic appointments, despite input from the diabetes outreach service. You called him to follow up. He agreed to have blood and urine tests then come to see you for a free 30-minute extended consultation, funded using your practice’s flexible funding scheme.
In the consult, you spend time re-establishing rapport with Mr V, who explains that he found hospital-based clinics difficult to attend due to unpredictable work commitments. However, he is worried about his health and highly motivated to make changes.
He and his family have recently started a church-based weight-loss and exercise programme. You congratulate him on this and offer support with monthly nurse weigh-ins and diet support.
You ask Mr V how else you can support him. He says he’s unsure why he takes all these medications when they don’t seem to work.
Can you suggest any changes?
Mr V is a good candidate for a medication review, given his suboptimal diabetes control on the current regimen.
What is empagliflozin?
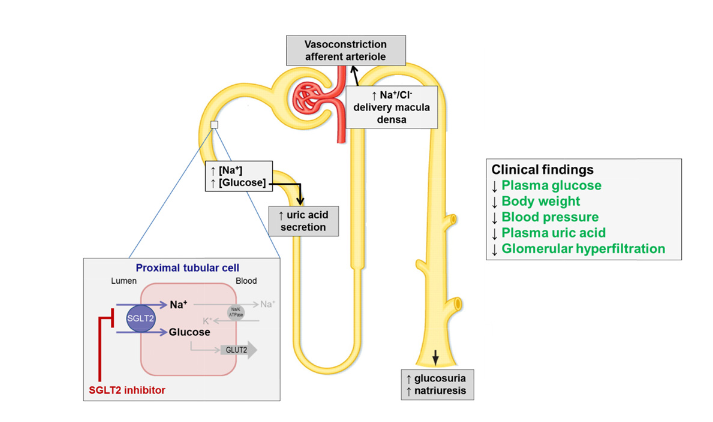
SGLT2 inhibitors (or ‘flozins’) work by getting rid of glucose and salt. They block the reabsorption of glucose and sodium in the proximal tubule, causing glucosuria and natriuresis (see diagram below). Clinically, expect reductions in:
- plasma glucose (mean reduction in HbA1c of 5 – 11 mmol/mol);
- blood pressure (mean reduction in systolic BP of 1 – 6 mmHg), and;
- body weight (mean reduction of 1 – 3 kg at 2 years).2,3
Mechanism of action of SGLT2 inhibitors.
(From SGLT2 inhibition in the diabetic kidney – from mechanisms to clinical outcome)
These effects translate into meaningful reductions in morbidity and mortality from diabetes-related renal and cardiovascular disease, as shown in the EMPA-REG trial and large meta-analyses.4
Notably, these benefits occur independently of the impact on glycaemic control.5
Importantly, although the glucose-lowering effects of SGLT2 inhibitors reduce with decreasing renal function, the benefits on renal and cardiovascular disease are maintained.
| Benefits of SGLT2 inhibitors in patients with type 2 diabetes |
|---|
NB: Reductions in mortality, atherosclerotic CVD and renal disease only occur in those patients with known disease (i.e. secondary prevention). There is no convincing evidence to date for SGLT2 inhibitors in primary prevention of CVD and/or renal disease. |
When should SGLT2 inhibitors be used?
SGLT2 inhibitors are recommended internationally and in New Zealand as second-line agents for the treatment of type 2 diabetes after metformin and lifestyle management. SGLT2 inhibitors are strongly recommended for all patients with type 2 diabetes with diabetic renal disease (urinary albumin/creatinine ratio > 3 mg/mmol and/or reduced eGFR) and/or heart failure and/or known CVD and/or a 5 year CVD risk > 15% irrespective of glycaemic control and other glucose-lowering therapies.
In all other patients with type 2 diabetes (i.e. those with no CVD or renal disease), SGLT2 inhibitors are useful second-line agents if inadequate glycaemic control, particularly in overweight or obese patients where weight loss is beneficial.
Empagliflozin is currently available on special authority6 for patients with type 2 diabetes with an HbA1c > 53 mmol/mol of Māori or Pacific ethnicity or at high risk of CVD or renal complications (see below for specific criteria).
It is important to know that Pharmac added the ethnicity criteria to reduce barriers to access and there is no evidence that SGLT2 inhibitors are more or less effective in Māori or Pacific peoples with type 2 diabetes. Furthermore, NZ guidelines5 recommend offering empagliflozin or dapagliflozin to patients who do not meet the current funding criteria. The estimated cost is around $85 per month.
What risks are associated with empagliflozin?
Hypotension due to volume depletion may occur with empagliflozin; take care in patients at risk for low blood pressure due to age (75 or older) or other medications, especially loop and thiazide diuretics. Rates of hypoglycaemia with empagliflozin monotherapy are similar to placebo, but there is a cumulative risk when empagliflozin is used with insulin or insulin secretagogues and it may be necessary to reduce doses of these medications when adding empagliflozin.3
Diabetic ketoacidosis (DKA)
A rare but serious adverse effect (1 in 3000 patients) of SGLT2 inhibitors is DKA. This presents with the usual symptoms and signs of DKA (nausea, vomiting, anorexia, abdominal pain, excessive thirst, difficulty breathing, confusion, unusual fatigue or sleepiness) but blood glucose levels may be normal or only slightly high (normoglycaemic DKA).
To prevent DKA, ask patients to stop their empagliflozin when they are unwell, or 2 days before an elective procedure, and restart the empagliflozin only once they are well again. Patients on empagliflozin who develop symptoms of DKA should be advised to attend your practice or hospital to ensure their capillary ketone levels are < 1.5 mmol/L.
Healthify has patient information on empagliflozin including DKA symptoms.
Increased risk of genitourinary infections, including Fournier’s gangrene
UTIs and other genital infections, including thrush or balanitis, can occur more frequently in patients taking empagliflozin. For recurrent infections, consider preventive treatment (such as Hiprex) or stop the empagliflozin. Fournier’s gangrene (necrotising fasciitis of the perineum) is rare but can have serious outcomes. Suspect this in patients with genital or perineal pain, erythema or swelling, particularly with associated fever or malaise.
When should empagliflozin NOT be used?
Empagliflozin is not recommended in patients with type 1 diabetes or diabetes from the loss of pancreatic function due to the risk of DKA, or in patients with stage 4 or 5 CKD (eGFR <30 mL/min/1.73m2) or on dialysis.
The risk of dehydration and hypotension means that empagliflozin is not recommended in patients aged 85 years and over, and caution is advised in those aged over 75 years. It is also not recommended in pregnancy, breastfeeding and in children < 18 years of age, though should be considered in patients diagnosed with type 2 diabetes in childhood given their high lifetime risk of CVD and diabetic renal disease.
Is empagliflozin suitable for Mr V?
Mr V has diabetic renal disease and his CVD risk is high based on his hypertension and ethnicity, so empagliflozin is a good choice. Together you review his latest test results:
- HbA1c 84 mmol/L
- Albumin/creatinine ratio (ACR) 54 mg/mmol
- eGFR 66 mL/min/1.73m2
- GGT 123 U/L; all other liver tests normal
- Lipids and FBC all within normal range
You explain to Mr V that there is a newly-available oral medication that will reduce glucose and salt in the body, and improve his diabetes along with the health of his heart and kidneys.
Mr V is open to trying it and wants to know more.
How to start empagliflozin
Before prescribing, discuss potential side effects and sick day management with particular attention to the increased risk of genitourinary infections and hypotension/dehydration, and the rare risks of DKA and Fournier’s gangrene.
Next, consider whether any other medications need to be stopped or reduced. SGLT2 inhibitors do not cause hypoglycaemia directly but improved glycaemic control in patients taking concomitant insulin and sulfonylureas may result in hypoglycaemia. Also consider stopping or reducing antihypertensives, especially diuretics, if the blood pressure is well controlled, to reduce the risk of hypotension.
Empagliflozin should be started at the lowest dose of 10mg daily and titrated up to 25mg daily as required.
No dosage adjustment is required for patients with hepatic impairment. A combination tablet with metformin may be useful to improve adherence.
Key points
- Empagliflozin can cause polyuria and volume depletion; consider reducing concomitant thiazide or loop diuretics, especially in older patients at risk of dehydration.
- Although empagliflozin itself doesn’t cause hypoglycaemia, the risk is increased with concomitant insulin or sulfonylureas:
- consider stopping or reducing doses of insulin by 15-20% and sulfonylureas by 50% if patients are close to target levels of glucose or HbA1c (e.g. < 64 mmol/mol).
- Urine will test positive for glucose while taking an SGLT2 inhibitor.
- Advise patients to stop their empagliflozin when unwell or 2 days before an elective procedure, then restarting the empagliflozin when well again.
- Consider stopping treatment if there are recurrent UTIs or other genitourinary infections.
You counsel Mr V about the expected glucose-lowering effect of empagliflozin, explaining that it could reduce his HbA1c by up to 11 mmol/mol. He will need further treatment to reach target HbA1c and you will continue to adjust the regimen over time.
You explain the potential side effects of empagliflozin and give him a patient handout. In discussing hypoglycaemia, you establish that he is aware of hypoglycaemic symptoms, but has had no episodes for over a year. Given the very high HbA1c you decide it is safe not to reduce the glipizide or insulin doses at this stage, but ask Mr V to monitor for hypoglycaemic symptoms and to measure his blood glucose before breakfast and before dinner for 3 days before he next sees you.
Next, you suggest that Mr V stops felodipine given that his blood pressure today is 118/78 mmHg; he is happy about this.
You ask if Mr V has any questions. He wonders whether he can take the combination tablet with metformin. You agree this is a good option and prescribe empagliflozin/metformin (Jardiamet) 5mg/1000mg tablets.
You ask Mr V to come back in 3 months after having a blood test so that you can review his HbA1c, home glucose readings, blood pressure and weight. In the meantime, you offer to have your nurse phone in a few weeks to check in; he agrees.
Finally, you give Mr V a written plan of the medication changes you have made today.
Practice points
- Empagliflozin (Jardiance) is recommended for the treatment of type 2 diabetes with cardiovascular disease (CVD) or high CVD risk, heart failure, or renal disease;
- Expect a 5-11 mmol/mol reduction in HbA1c, 1-6 mmHg reduction in systolic blood pressure, and 1-3kg reduction in weight at 2 years with empagliflozin;
- Consider reducing or stopping other diabetes or blood pressure medications if the patient is close to target;
- Consider proactively contacting patients who are eligible for funded treatment, including Māori and Pacific patients, and those with renal or cardiovascular complications
This MedCase was written by Dr Vicki Mount, BSC/BCom, MBChB, MRNZCGP, DipPaeds, with expert review by Dr Ryan Paul (BHB, MBChB, FRACP, PhD)
References
|
Pharmac Special Authority criteria for empagliflozin as at 1 February 20216 Empagliflozin is currently fully funded for patients with type 2 diabetes in New Zealand who are at high risk of cardiovascular or renal complications. Special authority criteria for fully funded empagliflozin include all of the following: 2. Any of the following: 3. Target HbA1c (of 53 mmol/mol or less) has not been achieved despite the regular use of at least one blood-glucose lowering agent (e.g. metformin, vildagliptin, or insulin) for at least 3 months; and 4. Treatment will not be used in combination with a funded GLP-1 agonist. Criteria 2.1 – 2.5 describe patients at high risk of cardiovascular or renal complications of diabetes. * Defined as: prior cardiovascular disease event (i.e. angina, myocardial infarction, percutaneous coronary intervention, coronary artery bypass grafting, transient ischaemic attack, ischaemic stroke, peripheral vascular disease), congestive heart failure or familial hypercholesterolaemia. ** Defined as: persistent albuminuria (albumin:creatinine ratio greater than or equal to 3 mg/mmol, in at least two out of three samples over a 3-6 month period) and/or eGFR less than 60 mL/min/1.73m2 in the presence of diabetes, without alternative cause. |
Recognition of Learning Activities
Don't forget to log your time with The Royal New Zealand College of General Practitioners portal for recognition of learning activities.


